9 0 5 7 8
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

氨磺必利 (Amisulpride) 对主要患有精神分裂症阴性症状的中国患者的疗效:ESCAPE 研究的分析
Authors Liang Y, Yu X
Received 2 May 2017
Accepted for publication 7 June 2017
Published 28 June 2017 Volume 2017:13 Pages 1703—1712
DOI https://doi.org/10.2147/NDT.S140905
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Prof. Dr. Roumen Kirov
Peer reviewer comments 2
Editor who approved publication: Professor Wai Kwong Tang
Background: Effective management strategies for the negative symptoms of
schizophrenia remain an unmet need, and data on the use of antipsychotics in
this population are scarce, particularly in Chinese patients. Therefore, we
investigated amisulpride for the treatment of Chinese patients with
predominantly negative symptoms of schizophrenia.
Methods: This post hoc subanalysis of the prospective
Effectiveness and Safety of Amisulpride in Chinese Patients with Schizophrenia
(ESCAPE) study included adult Chinese patients with an International
Classification of Diseases-10 diagnosis of schizophrenia and predominantly
negative symptoms, who received amisulpride for 8 weeks. Effectiveness outcomes
included ≥50% decrease in Positive and Negative Syndrome Scale (PANSS) negative
score, and a reduction in PANSS negative symptom score and Clinical Global
Impression Severity Scale (CGI-S). The study was registered at ClinicalTrials.gov (NCT01795183).
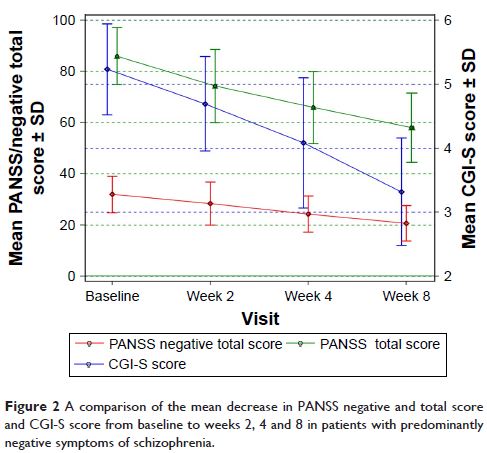
Results: In total, 26 patients were included in the analysis. A
≥50% decrease in PANSS negative score from baseline to week 8 was achieved by
34.6% of patients. From baseline to week 8, the mean PANSS negative
symptom score decreased by 45.2% (31.9 to 20.7) and CGI-S decreased
1.9 points (5.2 to 3.3). The mean week 8 dose of amisulpride was lower for
patients who achieved a ≥50% decrease in PANSS negative score at week 8 versus
those who did not (481.2 vs 704.1 mg/day). The most common treatment-related
adverse events included blood prolactin increase (19.2%) and extrapyramidal
disorder (19.2%). Weight gain was reported by one patient.
Conclusion: Amisulpride effectively reduced PANSS negative symptom
score and CGI-S for Chinese patients with predominantly negative symptoms of
schizophrenia. No unexpected adverse events were reported.
Keywords: schizophrenia,
amisulpride, negative symptoms, China