9 0 5 7 8
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

曲安奈德脂质纳米粒的经皮渗透性
Authors Qin ZM, Chen F, Chen DM, Wang Y, Tan YF, Ban JF
Received 5 December 2018
Accepted for publication 14 February 2019
Published 8 April 2019 Volume 2019:14 Pages 2485—2495
DOI https://doi.org/10.2147/IJN.S195769
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Govarthanan Muthusamy
Peer reviewer comments 3
Editor who approved publication: Dr Mian Wang
Background: Triamcinolone
acetonide (TAA) is an effective and the most commonly used corticosteroid
hormone for the treatment of hypertrophic scars (HSs). However, the clinically
used dosage has poor tissue permeability and injection safety. By contrast,
lipid nanoparticles (LNPs) have the advantage of high affinity for the skin.
Materials and methods: This
article describes the preparation of TAA-LNPs using poly(lactic-co-glycolic
acid) as a carrier material, which have good biocompatibility and
biodegradability. Based on a systematic investigation of its physicochemical
properties, a rabbit ear HSs model was established to evaluate the percutaneous
permeability of TAA-LNPs in scar tissue in vitro as well as to assess its
curative effect and skin irritation.
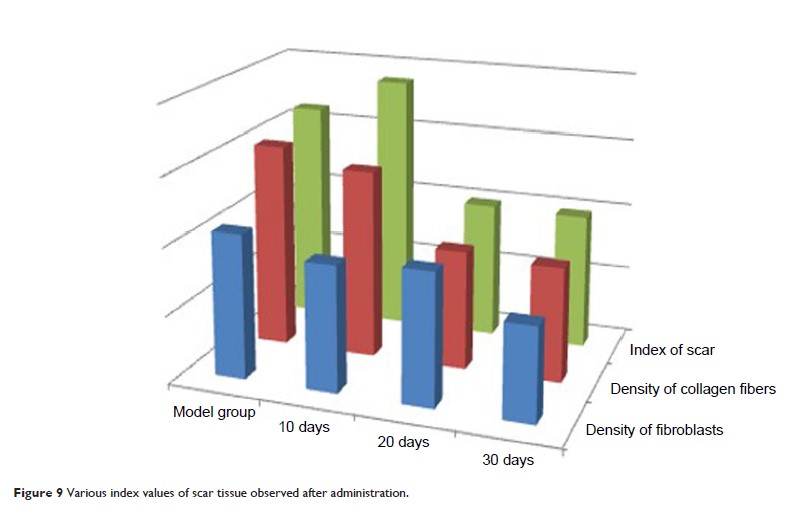
Results: The
results showed that the TAA-LNPs formed uniform and round particles under
fluoroscopy and had a complex structure in which a nanoparticle core was
surrounded by multiple vesicles. The particles were 232.2±8.2 nm in size,
and the complimentary potential was -42.16 mV. The encapsulation
efficiency was 85.24%, which is greater than that of other common liposomes and
nanoparticles. A test of in vitro scar tissue permeability showed that
penetration into scar tissue was twofold and 40-fold higher for TAA-LNPs than
for common liposome and commercial suspensions, respectively. The concentration
of the absorbed drug effectively inhibited fibroblast proliferation, achieved a
therapeutic effect in HSs, and did not stimulate intact or damaged skin.
Conclusion: The
preparation of TAA into LNPs for transdermal administration can enhance
transdermal permeation performance and the safety of this drug, which is
beneficial for the treatment of HSs.
Keywords: lipid
nanoparticles, transdermal permeation, triamcinolone acetonide, hypertrophic
scars