9 0 6 7 6
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

鞘内 TRPM8 阻断通过神经病理性疼痛大鼠背根神经节 PKC 和 NF-κB 信号通路减轻冷痛觉过敏
Authors Cao S, Li Q, Hou J, Li Z, Cao X, Liu X, Qin B
Received 5 December 2018
Accepted for publication 27 February 2019
Published 18 April 2019 Volume 2019:12 Pages 1287—1296
DOI https://doi.org/10.2147/JPR.S197168
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Andrew Yee
Peer reviewer comments 2
Editor who approved publication: Dr Katherine Hanlon
Background: TRPM8 channel plays central roles in the
sensitization of nociceptive transduction and is thought as one of the
potential targets for the treatment of neuropathic pain. However, the specific
molecular mechanisms are still less clear.
Methods: Sciatic
chronic constriction injury (CCI) rats were intrathecally administered with
AMTB (TRPM8-selective antagonist) or PDTC (nuclear factor-kappa B (NF-κB)
inhibitor). Cold-, thermal- and mechanical-pain thresholds were examined in CCI
and sham-operated rats before and after intrathecal administration of AMTB or
PDTC. Protein expression levels of TRPM8 and NF-κB p65, p-PKC/PKC value and
p-PKA/PKA value in the CCI ipsilateral L4-6 dorsal root ganglions (DRGs) were
analyzed. In addition, the co-expression of TRPM8 and NF-κB was evaluated in
DRG.
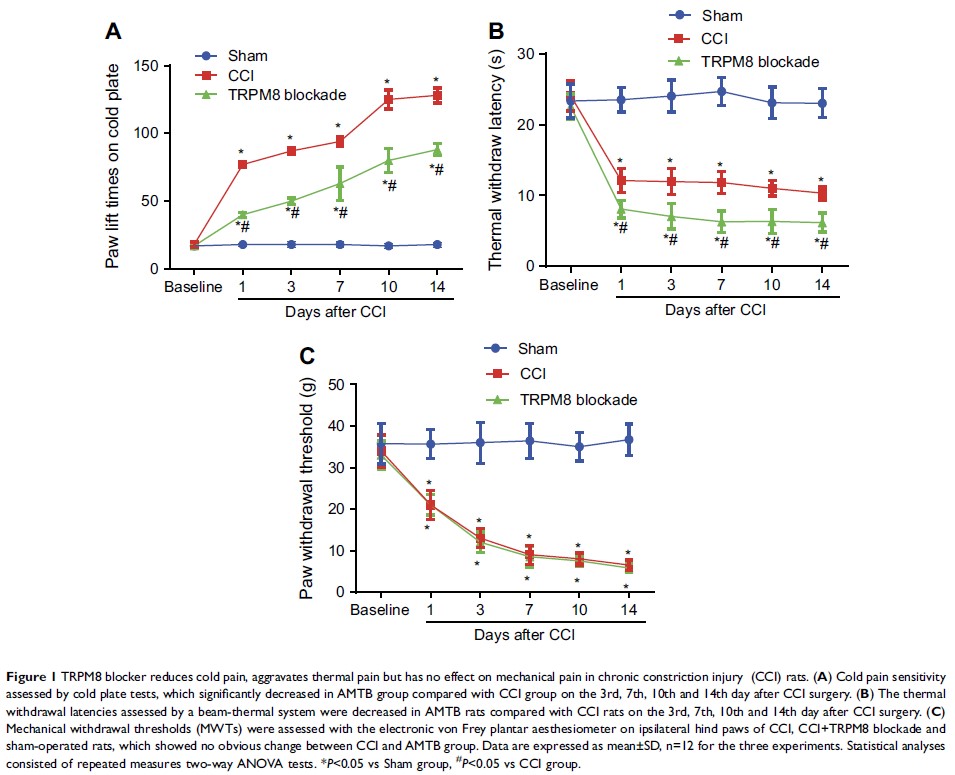
Results: Intrathecal
injection of AMTB decreased the cold hypersensitivity and aggravated the
thermal-hyperalgesia in the next 2 weeks after CCI surgery. The protein
expression of TRPM8 and NF-κB p65 in the ipsilateral DRGs significantly
increased after CCI surgery, which can be reversed by intrathecal
administration of AMTB. The PKC, PKA, p-PKC/PKC and p-PKA/PKA values showed
significantly increase after CCI surgery, while intrathecal AMTB administration
offset the expression increase of PKC, p-PKC and p-PKC/PKC but PKA or p-PKA/PKA
in the DRG. NF-κB inhibitor not only efficiently increased the cold-,
thermal-pain threshold of CCI rats, but also enhanced AMTB’s anti-cold pain
effect although exerted no anti-thermal hyperalgesia effect compared with TRPM8
blockade group. Immunofluorescence results showed co-expression of TRPM8 and
NF-κB in DRG neurons.
Conclusion: TRPM8
channels in DRGs participate in the pathogenesis of cold and thermal
hyperalgesia (not mechanical allodynia) in rats with neuropathic pain, which
could be regulated by PKC (not PKA) and NF-κB signaling. TRPM8 channel, PKC and
NF-κB are potential targets for cold hyperalgesia treatment in neuropathic pain
patients.
Keywords: TRPM8,
neuropathic pain, dorsal root ganglia, PKC, NF-κB