9 0 8 0 2
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

肽 SA12 通过 G0/G1 期的细胞周期阻滞抑制乳腺癌细胞系 MCF-7 和 MDA-MB-231 的增殖
Authors Yang LF, Liu HR, Long M, Wang X, Lin F, Gao ZW, Zhang HZ
Received 30 October 2017
Accepted for publication 17 January 2018
Published 30 April 2018 Volume 2018:11 Pages 2409—2417
DOI https://doi.org/10.2147/OTT.S154337
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Ru Chen
Peer reviewer comments 3
Editor who approved publication: Dr Samir Farghaly
Background: Targeted therapies have been proven as promising in the treatment of
breast cancer and have improved survival and quality of life in advanced breast
cancer. We previously identified a novel peptide SA12 which showed significant
activity in the inhibition of proliferation and induction of apoptosis in
SKBr-3 cells.
Methods: The present study investigated the potential antitumor role of
SA12 in breast cancer cell lines MDA-MB-231 and MCF-7 through Cell Counting
Kit-8 assay and colony formation assay, and examined the cell cycle
distribution using flow cytometry analysis. Furthermore, the expression of cell
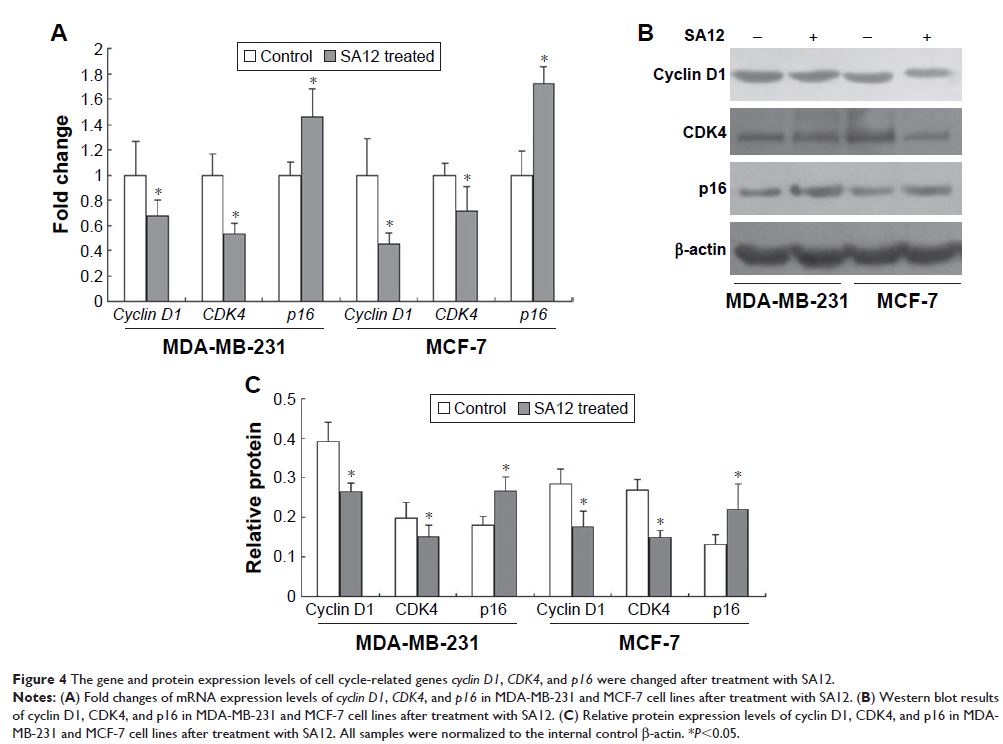
cycle-related genes cyclin D1 , CDK4 , and tumor suppressor
gene p16 were examined by
real-time polymerase chain reaction and Western blot to explore the molecular
mechanism.
Results: We determined that peptide SA12 suppressed the proliferation of
MDA-MB-231 and MCF-7 cell lines through the G0/G1 phase cell cycle arrest.
Moreover, the expressions of cell cycle-associated genes cyclin D1 and CDK4 were downregulated
and the expression of tumor suppressor gene p16 was
upregulated after treatment with SA12. MECP2 was required for the enhanced
expression of p16 gene
induced by SA12, which further inhibits CDK4/CDK6 activation and arrests the
cell cycle progression from G0/G1 to S phase.
Conclusion: We concluded that SA-12 inhibits the proliferation of MCF-7 and
MDA-MB-231 cells through G0/G1 cell cycle arrest. Cell cycle related
genes cyclin D1 , CDK4 , and p16 participate in the
process, and MECP2 is essential for the enhanced expression of p16 gene induced by SA-12.
Keywords: SA12, breast cancer, G0/G1 arrest, cyclin D1, CDK4, p16