9 0 8 0 2
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

阻止库容性 Ca2+ 内流可通过抑制在 Ca2+ 信号调节下生成 VEGF,来拮抗鼻咽癌中 Epstein-Barr 病毒所促进的血管生成
Authors Ye J, Huang J, He Q, Zhao W, Zhou X, Zhang Z, Li Y, Wei J, Zhang J
Received 10 December 2017
Accepted for publication 26 February 2018
Published 10 May 2018 Volume 2018:10 Pages 1115—1124
DOI https://doi.org/10.2147/CMAR.S159441
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Andrew Yee
Peer reviewer comments 2
Editor who approved publication: Professor Kenan Onel
Background: Epstein–Barr virus (EBV) actively contributes to the pathological
process of nasopharyngeal carcinoma (NPC) by enabling NPC cells to acquire
various capacities required for their malignant biological actions. Our earlier
works demonstrated that EBV-encoded latent membrane protein 1 (LMP1) enhanced
vascular endothelial growth factor (VEGF)-mediated angiogenesis by boosting
store-operated Ca2+ entry (SOCE) upon
extracellular epidermal growth factor (EGF) stimulation. However, the
antagonistic effects of SOCE blockage on EBV-promoted angiogenesis must be
appropriately evaluated in vivo, and the global effect of EBV infection on the
EGF-elicited cytosolic Ca2+ signaling,
which regulates VEGF-mediated angiogenesis remains to be further
clarified.
Materials and methods: Two EBV-infected NPC cell lines, CNE2-EBV and
HK1-EBV, along with their parental cell lines were employed in the present
study. Dynamic cytosolic Ca2+ changes
were measured in individual fluorescent Ca2+ indicator-loaded
cells. Amounts of VEGF production were determined by enzyme-linked
immunosorbent assay (ELISA). Human umbilical vein endothelial cells
(HUVECs)-formed tube networks were quantitatively evaluated as an in vitro
angiogenesis assay. A mouse model concurrently bearing EBV-positive/negative
xenografts was utilized to evaluate the tumor growth and angiogenesis in vivo.
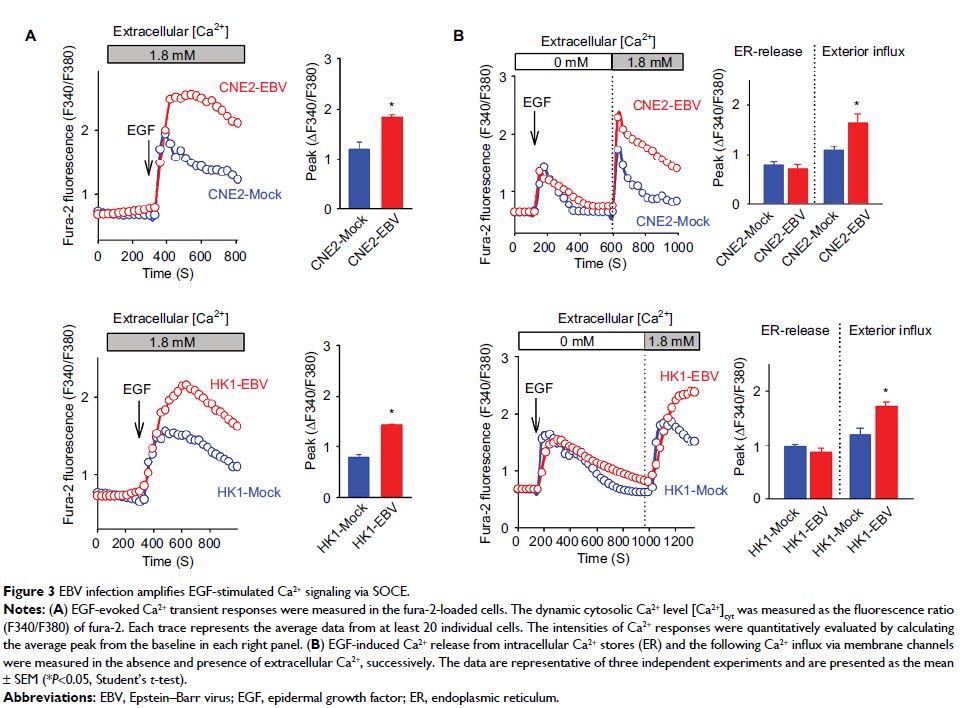
Results: EBV infection reliably promoted transplanted
tumor growth while enhancing angiogenesis. Introduction of EBV into
EBV-negative NPC cells increased the EGF-stimulated VEGF production while
amplifying the EGF-evoked Ca2+ responses.
Inhibition of the EBV-boosted Ca2+ signaling
using 2-aminoethyl diphenylborinate (2-APB), a specific SOCE inhibitor,
effectively antagonized the EBV-promoted VEGF production and endothelial tube
formation in vitro. Pharmacological blockage of SOCE exhibited anti-angiogenic
effect in the EBV-positive xenografts.
Conclusion: SOCE can serve as a candidate pharmacological
target for treating NPC, as blockage of the Ca2+ signaling
via SOCE is a feasible strategy to suppress the EBV-driven malignant profiles
in NPC cells.
Keywords: nasopharyngeal
carcinoma, store-operated Ca2+ entry,
angiogenesis, Epstein–Barr virus, vascular endothelial growth factor