9 0 8 1 0
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

上调的清道夫受体 CD36 与动脉粥样硬化兔模型支架置入后非靶区病变的进展相关
Authors Li R, Cui S, Xu Y, Xing J, Xue L, Chen Y
Received 11 July 2018
Accepted for publication 26 September 2018
Published 13 November 2018 Volume 2018:11 Pages 447—456
DOI https://doi.org/10.2147/JIR.S179814
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Amy Norman
Peer reviewer comments 2
Editor who approved publication: Dr Ning Quan
Background: The
incidence of recurrent cardiovascular events from the progression of nontarget
lesions (NTLs) is high for percutaneous coronary intervention-treated patients.
However, the underlying mechanisms have not been thoroughly elucidated.
Methods: In this
study, ten atherosclerotic rabbits with multiple plaques in the upper and lower
segments of abdominal aorta (group A) were randomly divided into two subgroups:
group A1 underwent intravascular ultrasound examination and stent implantation
in the lower segments of the abdominal aorta (n=5), whereas group A2 was
without stenting (n=5). Group B was a control group without balloon injury. The
serum levels of high-sensitivity CRP, interleukin-6 (IL-6), oxidized
low-density lipoprotein, and CD36 were assessed via ELISA at five time points
between the 10th and 18th weeks. The upper abdominal aorta was examined via the
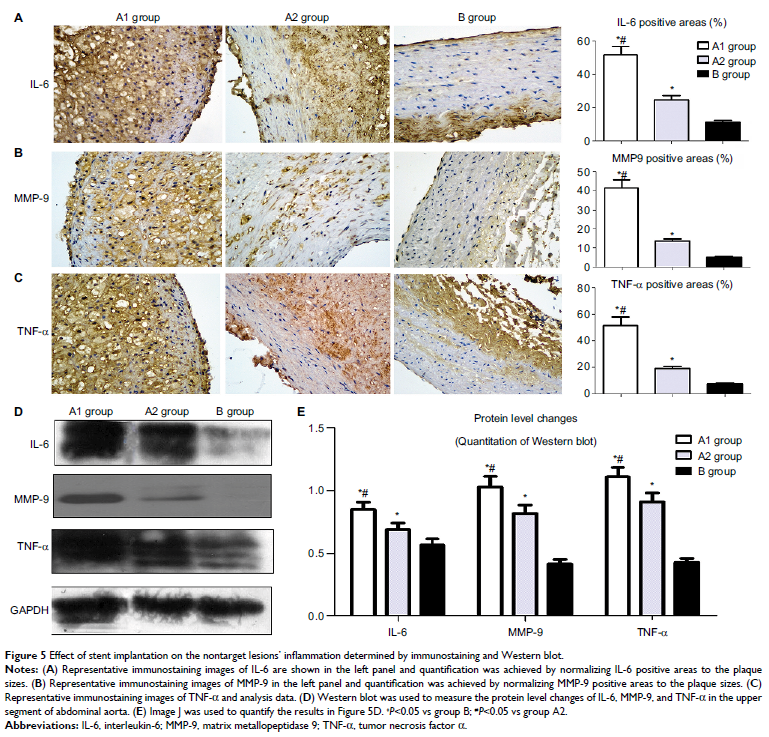
immunohistochemical stain and Western blotting of matrix metallopeptidase 9
(MMP-9), CD36, IL-6, and tumor necrosis factor α.
Results: As a
result, we found that stent implantation aggravated serum levels of CD36,
oxidative stress, and inflammatory cytokines. Meanwhile, the upper abdominal
arterial plaque burden significantly increased after stenting by intravascular
ultrasound. Immunohistochemistry and Western blotting showed that the local NTLs’
matrix metallopeptidase 9, CD36, IL-6, and tumor necrosis factor α expressions
in group A1 were significantly higher than those in groups A2 and B (P <0.05–0.01).
More importantly, a strong correlation was identified between CD36 expression
and NTLs’ plaque burden before the rabbits were killed.
Conclusion: Taken
together, stent implantation accelerated inflammation, induced oxidative
stress, and increased the NTLs’ progression, which were associated with the
upregulated CD36 expression.
Keywords: inflammation,
oxidative stress, CD36, atherosclerosis, stent