9 0 8 0 2
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

PNUT 通过 PI3K/AKT 信号通路介导电离辐射诱导的 CNE-2 鼻咽癌细胞迁移、侵袭和上皮 - 间质转化
Authors Yu D, An X, Fan W, Wang X, He Y, Li B
Received 9 October 2018
Accepted for publication 14 January 2019
Published 15 February 2019 Volume 2019:12 Pages 1205—1214
DOI https://doi.org/10.2147/OTT.S188571
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Justinn Cochran
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Geoffrey Pietersz
Purpose: Ionizing
radiation (IR) is widely used for treating nasopharyngeal carcinoma (NPC).
However, recent studies indicate that IR can also promote the migration and
invasion of malignant tumors. Phosphatase 1 nuclear-targeting subunit (PNUTS),
a novel interacting protein, was recently demonstrated to be involved in
tumorigenesis and metastasis formation. This protein was hypothesized to take
part in IR-induced migration and invasion in NPC cells in this study.
Materials and methods: Western
blotting was used to detect how PNUTS was expressed in NPC cells with or
without IR treatment. Wound-healing and Transwell assays were used to measure
cell migration and invasion. Quantitative real-time PCR and Western blotting
were used to determine the expression levels of PNUTS and
epithelial–mesenchymal transition (EMT) proteins, respectively, after CNE-2
cells were infected with an adenovirus vector, ad-PNUTS, or transfected with
PNUTS-specific siRNA. Finally, the expression levels of PI3K/AKT
signaling-related proteins were detected by Western blotting.
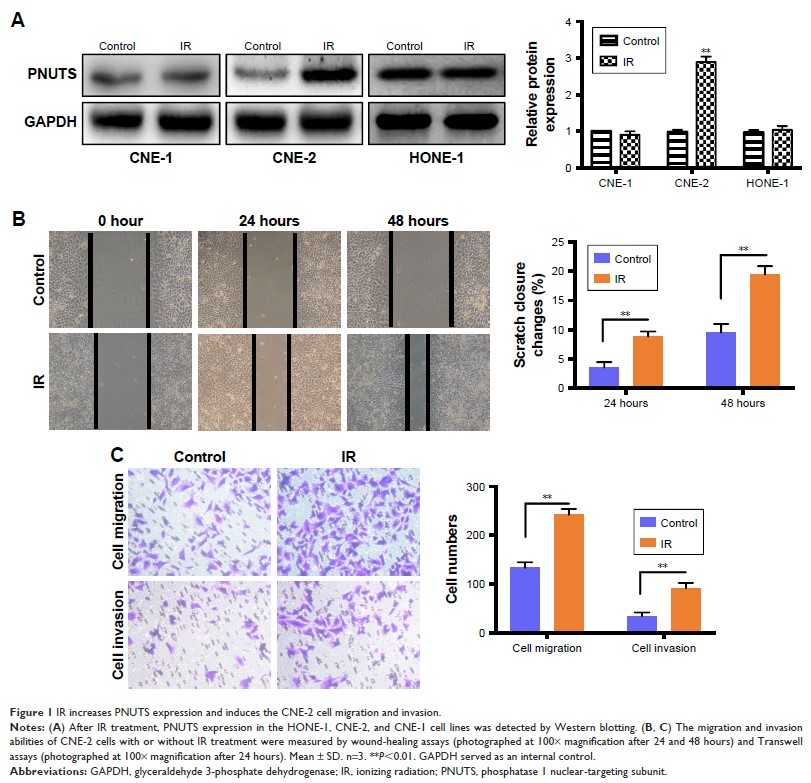
Results: IR
significantly promoted PNUTS expression and the migration and invasion in CNE-2
cells. Moreover, after exposure to IR, expression of the mesenchymal markers
N-cadherin and vimentin increased, while that of the epithelial marker
E-cadherin decreased. Silencing PNUTS remarkably attenuated IR-induced
increases in cell migration and invasion and reversed the EMT process.
Additionally, the overexpression of PNUTS restored the mobility and
invasiveness of CNE-2 cells, which regained EMT characteristics. Furthermore,
we found that PNUTS regulated IR-induced EMT via the PI3K/AKT signaling
pathway.
Conclusion: Our
research illustrates a relationship between PNUTS and IR-induced cell migration
and invasion and provides a novel therapeutic target for preventing
radiotherapy-induced metastasis in NPC patients.
Keywords: PNUTS,
ionizing radiation, EMT, PI3K/AKT pathway, NPC