9 0 8 0 2
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

已发表论文
通过阻断转化生长因子-β 信号通路增强 CIK 细胞过继性治疗对乳腺癌的体外抗肿瘤效应
Authors Zhao Y, Hu JY, Li RG, Song J, Kang YJ, Liu S, Zhang DW
Published Date June 2015 Volume 2015:8 Pages 1553—1559
DOI http://dx.doi.org/10.2147/OTT.S82616
Received 11 February 2015, Accepted 31 March 2015, Published 22 June 2015
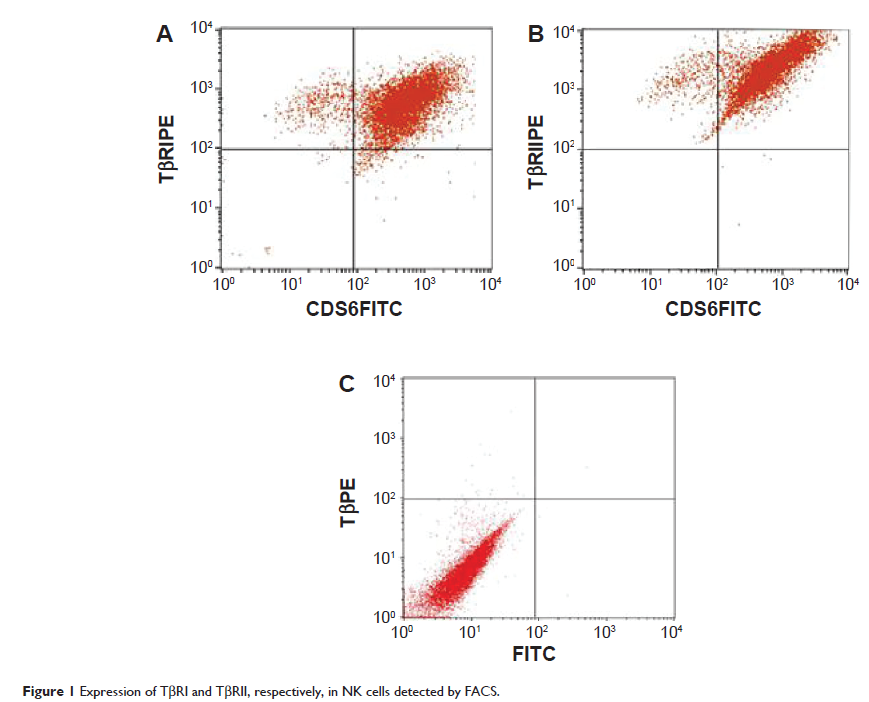
Abstract: Natural killer (NK) cells have great potential for improving cancer immunotherapy. Adoptive NK cell transfer, an adoptive immunotherapy, represents a promising nontoxic anticancer therapy. However, existing data indicate that tumor cells can effectively escape NK cell-mediated apoptosis through immunosuppressive effects in the tumor microenvironment, and the therapeutic activity of adoptive NK cell transfer is not as efficient as anticipated. Transforming growth factor-beta (TGF-β) is a potent immunosuppressant. Genetic and epigenetic events that occur during mammary tumorigenesis circumvent the tumor-suppressing activity of TGF-β, thereby permitting late-stage breast cancer cells to acquire an invasive and metastatic phenotype in response to TGF-β. To block the TGF-β signaling pathway, NK cells were genetically modified with a dominant-negative TGF-β type II receptor by optimizing electroporation using the Amaxa Nucleofector system. These genetically modified NK cells were insensitive to TGF-β and resisted the suppressive effect of TGF-β on MCF-7 breast cancer cells in vitro. Our results demonstrate that blocking the TGF-β signaling pathway to modulate the tumor microenvironment can improve the antitumor activity of adoptive NK cells in vitro, thereby providing a new rationale for the treatment of breast cancer.
Keywords: transforming growth factor-beta, natural killer cells, breast cancer, adoptive immunotherapy
Keywords: transforming growth factor-beta, natural killer cells, breast cancer, adoptive immunotherapy