9 1 2 3 6
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

尼古丁通过调节小鼠的过氧化物酶 1 和上皮 - 间质转化来促进口腔癌的颈部转移
Authors Wang M, Niu W, Qi M, Chen H, Zhang M, Wang C, Ge L, Yang J, Miao C, Shi N, Chen T, Tang X
Received 10 November 2018
Accepted for publication 13 February 2019
Published 1 May 2019 Volume 2019:12 Pages 3327—3338
DOI https://doi.org/10.2147/OTT.S194129
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Andrew Yee
Peer reviewer comments 2
Editor who approved publication: Dr Federico Perche
Background: Tobacco
is a major risk factor for oral squamous cell carcinoma (OSCC). However, the
role of nicotine in OSCC is not completely understood.
Materials and methods: To
analyze the mechanisms of nicotine-induced cervical metastasis, we investigated
whether nicotine induced invasion, migration, and epithelial–mesenchymal
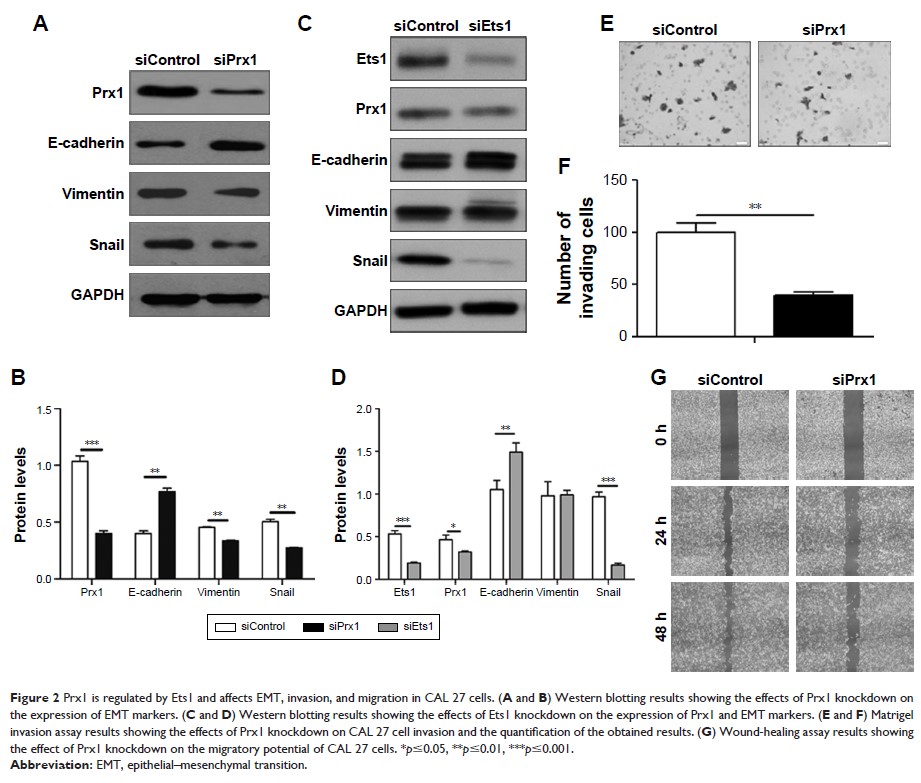
transition (EMT) via regulating peroxiredoxin 1 (Prx1) in CAL 27 cells. In
addition, we established a mouse model to confirm the roles of nicotine in
regulating Ets1/Prx1/EMT signaling in OSCC metastasis.
Results: We showed
that nicotine induced CAL 27 cell invasion, migration, EMT, and Prx1 and Ets1
expression. Prx1 knockdown inhibited cell invasion, migration, and EMT. Ets1
silencing downregulated Prx1 expression and EMT. Prx1 and Ets1 were shown to
interact in CAL 27 cells treated with nicotine, and nicotine could
significantly upregulate the binding of the transcription factor Ets1 to the
Prx1 gene promoter region. Additionally, an in vivo study showed that
nicotine induced tumor metastasis and EMT. Prx1 knockdown inhibited cervical
metastasis rates and EMT progression. No significant differences in metastasis
rates and EMT-related marker expression levels were observed between vehicle-
and nicotine-treated mice.
Conclusion: The
results indicate that nicotine promotes cervical lymph node metastasis through
regulating Ets1/Prx1/EMT signaling during OSCC pathogenesis; consequently, Prx1
may represent a potential target for the prevention and treatment of OSCC.
Keywords: oral
squamous cell carcinoma, peroxiredoxin 1, metastasis, epithelial–mesenchymal
transition, mouse model