9 0 8 0 2
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 2.6 Breast Cancer (Dove Med Press)
- 3.9 Clin Epidemiol
- 3.3 Cancer Manag Res
- 3.9 Infect Drug Resist
- 3.6 Clin Interv Aging
- 4.8 Drug Des Dev Ther
- 2.8 Int J Chronic Obstr
- 8.0 Int J Nanomed
- 2.3 Int J Women's Health
- 3.2 Neuropsych Dis Treat
- 4.0 OncoTargets Ther
- 2.2 Patient Prefer Adher
- 2.8 Ther Clin Risk Manag
- 2.7 J Pain Res
- 3.3 Diabet Metab Synd Ob
- 4.3 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.9 Pharmgenomics Pers Med
- 3.5 Risk Manag Healthc Policy
- 4.5 J Inflamm Res
- 2.3 Int J Gen Med
- 4.1 J Hepatocell Carcinoma
- 3.2 J Asthma Allergy
- 2.3 Clin Cosmet Investig Dermatol
- 3.3 J Multidiscip Healthc

一项新的 LARCassigner3 分类预测了新辅助化放疗治疗局部晚期直肠癌患者的预后: 回顾性培训和验证分析
Authors Zhang J, Shen L, Deng Y, Sun X, Wang Y, Yao Y, Zhang H, Zou W, Zhang Z, Wan J, Yang L, Zhu J, Zhang Z
Received 1 December 2018
Accepted for publication 19 March 2019
Published 7 May 2019 Volume 2019:11 Pages 4153—4170
DOI https://doi.org/10.2147/CMAR.S196662
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Andrew Yee
Peer reviewer comments 3
Editor who approved publication: Dr Chien-Feng Li
Purpose: To build
and validate a predictive model of outcome for patients with locally advanced
rectal cancer (LARC) treated with neoadjuvant chemoradiotherapy.
Materials and methods: We
developed a LARCassigner3 classifier based on tumor and paired normal tissues
of patients treated with neoadjuvant chemoradiation and surgery from January
2007 to December 2012 in Fudan University Shanghai Cancer Center. Excluding 23
pairs of tissues failed in the RNA quality test, rested 197 patients were
divided into discovery (n=98) and validation (n=99) cohorts randomly. Median
follow-up time was 58 months. We used the Kaplan–Meier method to estimate
disease-free survival (DFS), overall survival (OS), local recurrent, and distant
metastatic rate We constructed a multivariate Cox model to identify the
variables independently associated with progression-free and OS.
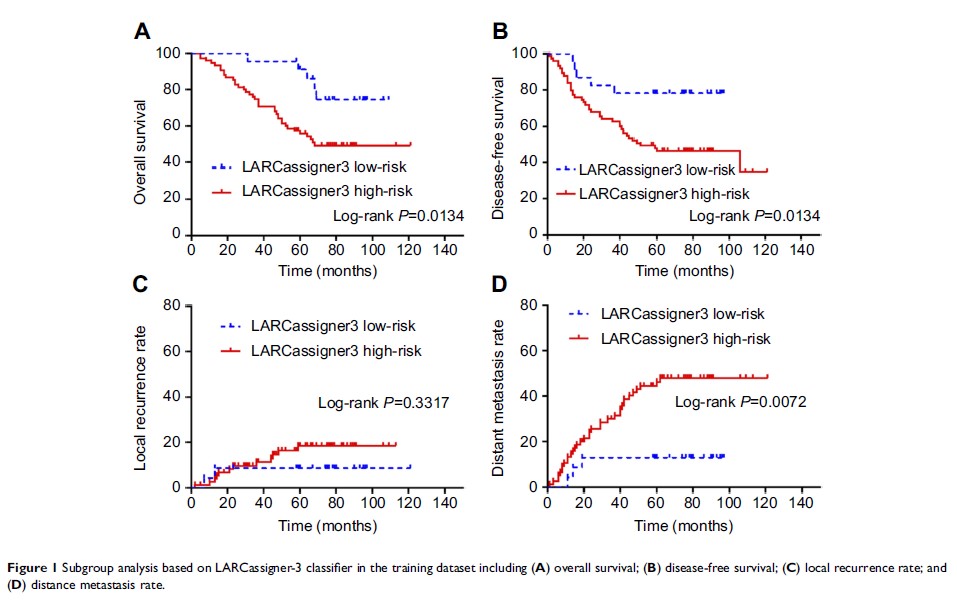
Results: We
identified three classifier genes related to relevant colorectal cancer
features (CXCL9, SFRP2, and CD44) that formed the LARCassigner3 classifier
assay. In the discovery set, the median DFS was 48.1 months (95% confidence
interval (CI) 47.3–49.5) in the low-risk group and 23.4 months (95% CI
22.1–24.8) in the high-risk group (p =0.0134); the median OS was 39.2 months (95% CI
38.4–40.3) in the high-risk group and 19.1 months (95% CI 18.3–20.7) in the
low-risk group (p =0.0134);
5-year distant metastasis was 13.9% (95% CI 9.0–21.3) in the low-risk group and
49.8% (95% CI 38.7–60.9) in the high-risk group (p =0.0072).
Additionally, the different responses to neoadjuvant chemoradiotherapy and the
LARCassigner3 low-risk and high-risk groups was statistically significant (p =0.004) in the
discovery cohort. Similar results were obtained in the internal evaluation
cohort.
Conclusions: Patients
with LARCassigner3 low-risk tumors were associated with a good prognosis. The
clinical utility of using LARCassigner3 subtyping for the identification of
patients for neoadjuvant chemoradiotherapy requires validation in dependent clinical
trial cohorts.
Keywords: prognostic
marker, locally advanced rectal cancer, neoadjuvant chemoradiotherapy