113173
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

非小细胞肺癌患者 PD-L1 表达的检测及其在循环肿瘤细胞中的临床意义
Authors Cheng Y, Wang T, Lv X, Li R, Yuan L, Shen J, Li Y, Yan T, Liu B, Wang L
Received 9 January 2020
Accepted for publication 27 February 2020
Published 19 March 2020 Volume 2020:12 Pages 2069—2078
DOI https://doi.org/10.2147/CMAR.S245425
Checked for plagiarism Yes
Review by Single-blind
Peer reviewer comments 2
Editor who approved publication: Dr Antonella D'Anneo
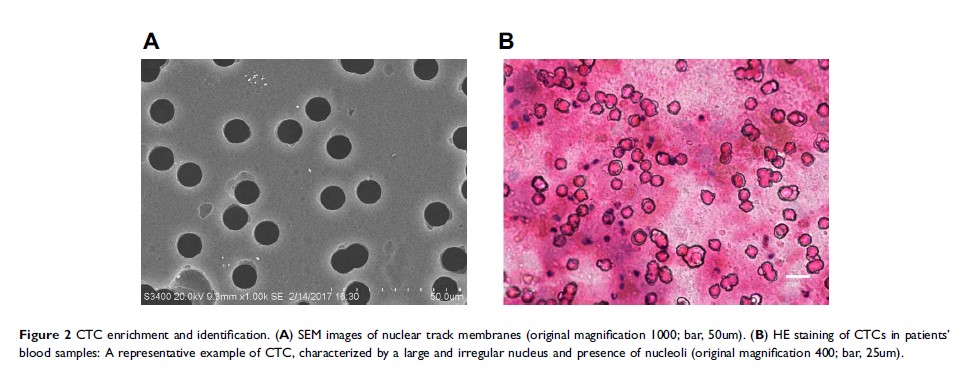
Background: The expression of programmed cell death ligand 1(PD-L1) is related to the efficacy of immune checkpoint inhibitors on patients with non-small cell lung cancer (NSCLC), but tumor tissue (TT) samples are difficult to obtain, and initial TT samples are difficult to reflect the spatial-temporal heterogeneity. Therefore, we explored the feasibility of separating circulating tumor cells (CTCs) and detecting PD-L1 expression on CTCs.
Patients and Methods: Peripheral blood specimens were sampled from 66 NSCLC patients, and CTCs were separated by membrane filtration based on size. For 59 patients with paired TT specimens, the expression of PD-L1 in their CTCs and TTs was determined using the immunohistochemistry and immunocytochemistry based on 28– 8 antibody, respectively. The PD-L1 expression in TTs was set as a gold standard for calculation of sensitivity, specificity, consistency, positive predictive value (PPV), and negative predictive value (NPV), and the Cohen kappa coefficient for CTCs and paired TTs was calculated. In addition, the T-test, Chi-square test, and Mann–Whitney U-test were adopted to analyze the correlation of clinical pathological features and prognosis with PD-L1 expression.
Results: Sensitivity, specificity, concordance, PPV and NPV of detecting PD-L1 in CTCs of the 41 initial treated patients were 88.89%, 73.91%, 80%, 72.73% and 89.47%, respectively, and the Cohen kappa coefficient of CTC and paired TTs was 0.613. The univariate analysis of survival showed that the progression-free survival time of initial treated patients with positive PD-L1 expression was shorter than that of those with negative PD-L1 expression in CTCs or TTs (P > 0.05), and the positive PD-L1 expression in CTCs or TTs had nothing to do with age, sex, smoking status, histological type, and stage (P > 0.05).
Conclusion: The study confirms the feasibility of CTC PD-L1 detection in peripheral blood and lays a foundation for exploring real-time and individualized immunotherapy molecular biomarkers.
Keywords: circulating tumor cell, non-small cell lung cancer, immunotherapy, PD-L1 level/expression