113173
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

ARID1A 的缺失通过 NPC 中的 Akt 信号通路促进增殖、迁移和侵袭
Authors Yang Y, Wang X, Yang J, Duan J, Wu Z, Yang F, Zhang X, Xiao S
Received 1 March 2019
Accepted for publication 10 May 2019
Published 29 May 2019 Volume 2019:11 Pages 4931—4946
DOI https://doi.org/10.2147/CMAR.S207329
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Cristina Weinberg
Peer reviewer comments 2
Editor who approved publication: Dr Alexandra R. Fernandes
Background: AT-rich interactive domain-containing protein 1A (ARID1A) is a member of the switch/sucrose nonfermentable chromatin remodeling complex, which has been observed to be mutated in various tumors. The loss of ARID1A is reported to be frequently associated with PI3K/Akt pathway activation.
Objective: The roles of ARID1A in nasopharyngeal carcinoma (NPC) have not been reported until now. The aim of this research was to explore the clinical significance and potential mechanism of ARID1A in NPC development and progression.
Methods: ARID1A expression levels were investigated in human NPC tissues and cell lines. The effects of ARID1A knockdown on nasopharyngeal cancer cell proliferation, migration and invasion were evaluated in vitro using CCK8, wound healing, transwell and flow cytometry assays. The expression of relevant proteins was evaluated by Western blot assays.
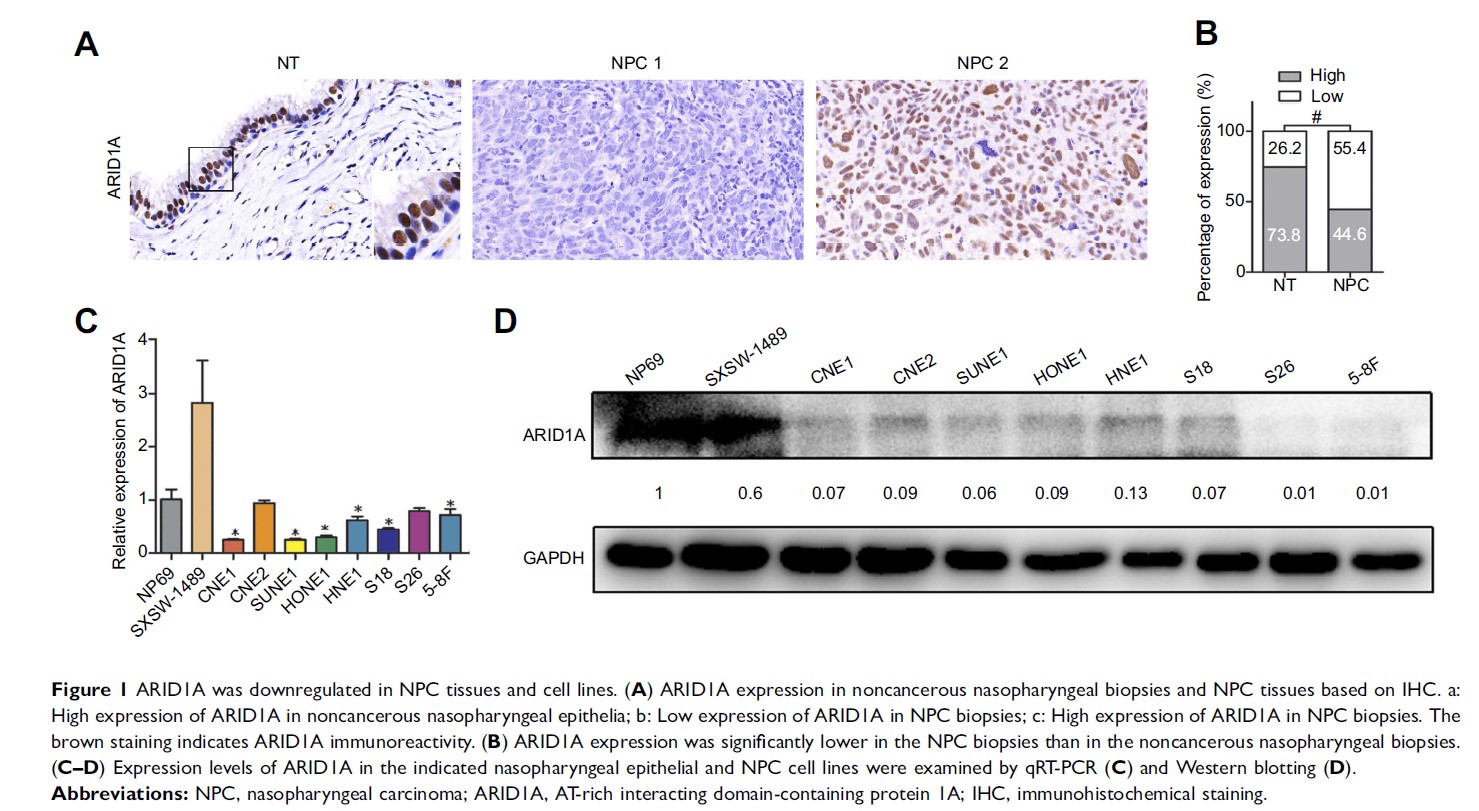
Results: In this study, ARID1A was significantly downregulated in NPC tissues and cells. Furthermore, low ARID1A expression was significantly associated with aggressive clinicopathological characteristics and poor survival in NPC patients. Depletion of endogenous ARID1A by siRNA promoted proliferation, migration and invasion in CNE1 and HNE1 cells. Additionally, ARID1A knockdown increased the phosphorylation of Akt in NPC cells. High levels of p-Akt were also observed in NPC biopsies and correlated with ARID1A downregulation. These results imply that the loss of ARID1A could activate Akt signaling. In addition, MK-2206 (a highly selective inhibitor of Akt) partially suppressed NPC cell proliferation, migration and invasion, which were induced by ARID1A knockdown.
Conclusion: Our findings indicate that ARID1A plays an essential role in modulating the Akt pathway, functions as a tumor suppressor in NPC and may be a potential target for NPC treatment.
Keywords: nasopharyngeal carcinoma, SWI/SNF, ARID1A, PI3K/Akt pathway, Akt inhibitor