113173
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

开发用于长春新碱的稳定单瓶脂质体制剂
Authors Mao W, Wu F, Lee RJ, Lu W, Wang J
Received 13 February 2019
Accepted for publication 8 May 2019
Published 21 June 2019 Volume 2019:14 Pages 4461—4474
DOI https://doi.org/10.2147/IJN.S205276
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Eytan Klausner
Peer reviewer comments 2
Editor who approved publication: Dr Linlin Sun
Background: Vincristine is a potent therapeutic agent with well-defined activity against hematologic malignancies and solid tumors. It is a cell-cycle specific drug with concentration and exposure duration dependent activity. When used by liposomal delivery, it exhibits enhanced anti-tumor activity. However, vincristine liposome formulation in the clinic is supplied as a 3-vial-kit due to lacking sufficient stability. So it has to be prepared in situ prior to use through a multi-step process.
Purpose: The purpose here is to develop a more stable and ready-to-use liposomal formulation for vincritstine in one vial.
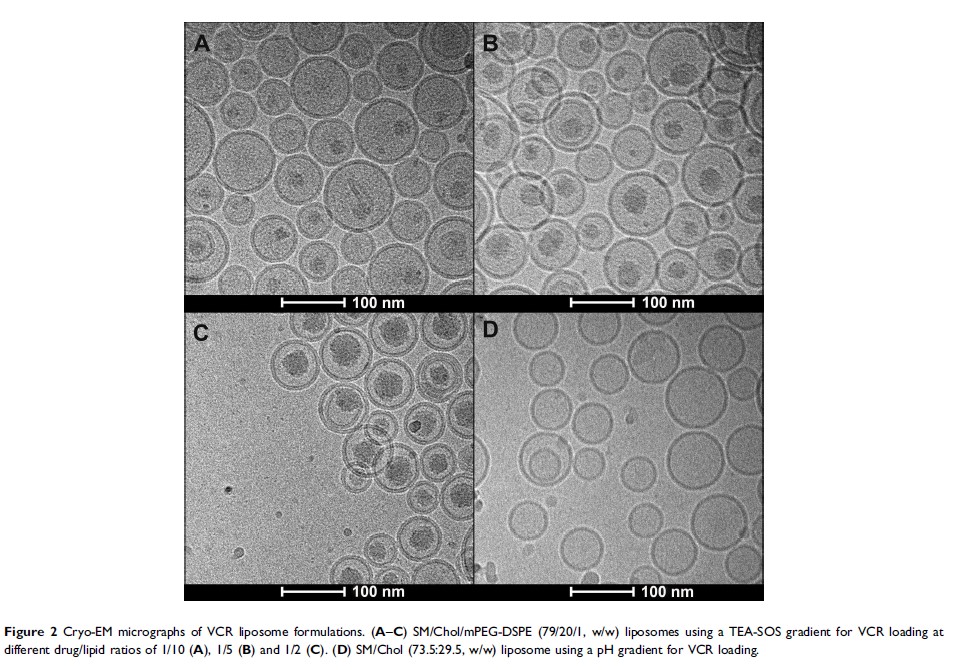
Patients and methods: A series of preparations were investigated based on sphingomyelin/cholesterol/PEG2000-DSPE lipid composition, with different drug/lipid (D/L) ratios (1/10, 1/5, 1/2), using an active sucrose octasulfate triethylamine salt gradient loading method. In this work, compared to generic vincristine sulfate liposome injection (GVM), the stability both in vivo and in vitro and efficacy in vivo of novel vincristine liposomes were investigated.
Results: It was shown that the degradation of vincristine during 2–8°C storage was significantly decreased from 8.2% in 1 month (GVM) to 2.9% in 12 months (D/L ratio 1/5). The half-time for sphingomyelin/cholesterol/PEG2000-DSPE liposomes in vivo could be adjusted from 17.4 h (D/L ratio 1/10) to 22.7 h (D/L ratio 1/2) in rats, while the half-time for GVM was only 11.1 h. The increase in drug retention contributed to the lower in vivo toxicity. The antitumor efficacy was evaluated using a human melanoma tumor model and showed remarkable improvement compared to GVM.
Conclusion: The study demonstrates that the new formulation with the drug/lipid ratio of 1/5 owns a higher encapsulation efficiency, better stability, lower toxicity and superior antitumor efficacy, which is screened out for further development.
Keywords: vincristine sulfate, liposome, TEA-SOS, storage stability, and anti-tumor efficacy