113173
论文已发表
注册即可获取德孚的最新动态
IF 收录期刊
- 3.4 Breast Cancer (Dove Med Press)
- 3.2 Clin Epidemiol
- 2.6 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.7 Clin Interv Aging
- 5.1 Drug Des Dev Ther
- 3.1 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.6 Int J Women's Health
- 2.9 Neuropsych Dis Treat
- 2.8 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.2 Ther Clin Risk Manag
- 2.5 J Pain Res
- 3.0 Diabet Metab Synd Ob
- 3.2 Psychol Res Behav Ma
- 3.4 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.0 Risk Manag Healthc Policy
- 4.1 J Inflamm Res
- 2.0 Int J Gen Med
- 3.4 J Hepatocell Carcinoma
- 3.0 J Asthma Allergy
- 2.2 Clin Cosmet Investig Dermatol
- 2.4 J Multidiscip Healthc

痰液外泌体微 RNA 分析揭示了在支气管扩张中由铜绿假单胞菌定植所调控的关键信号通路
Authors Huang Y, Chen C, Yuan J, Li H, Han X, Chen R, Guan W, Zhong N
Received 17 June 2019
Accepted for publication 21 October 2019
Published 22 November 2019 Volume 2019:14 Pages 2563—2573
DOI https://doi.org/10.2147/COPD.S219821
Checked for plagiarism Yes
Review by Single-blind
Peer reviewers approved by Dr Cristina Weinberg
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Chunxue Bai
Background: Pseudomonas aeruginosa (PA) colonization confers poor prognosis in bronchiectasis. However, the biomarkers and biological pathways underlying these associations are unclear.
Objective: To identify the roles of PA colonization in bronchiectasis by exploring for sputum exosomal microRNA profiles.
Methods: We enrolled 98 patients with clinically stable bronchiectasis and 17 healthy subjects. Sputum was split for bacterial culture and exosomal microRNA sequencing, followed by validation with quantitative polymerase chain reaction. Bronchiectasis patients were stratified into PA and non-PA colonization groups based on sputum culture findings. We applied Gene Ontology and Kyoto Encyclopedia of Genes and Genome pathway enrichment analysis to explore biological pathways corresponding to the differentially expressed microRNAs (DEMs) associated with PA colonization.
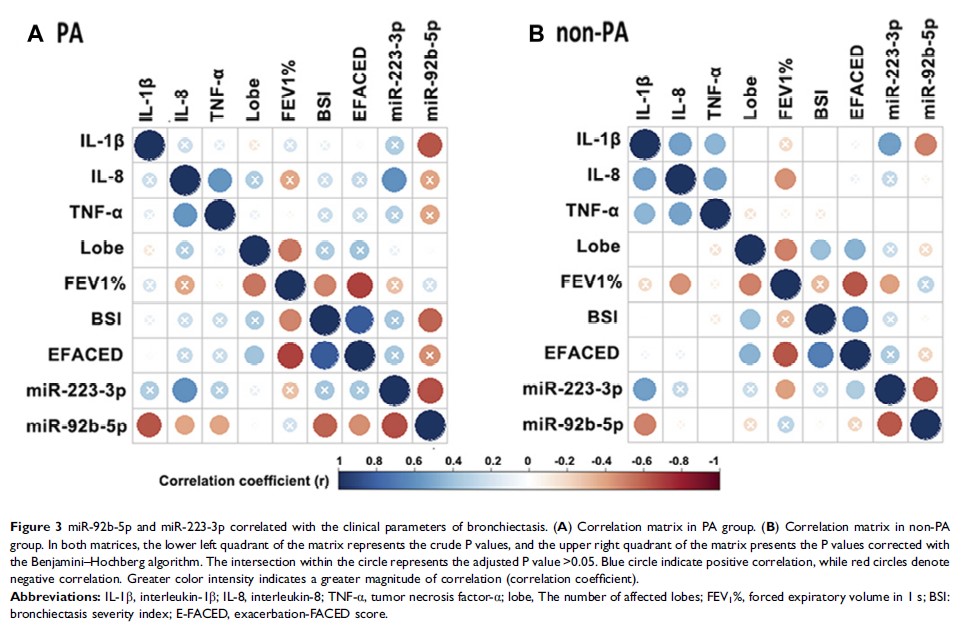
Results: Eighty-two bronchiectasis patients and 9 healthy subjects yielded sufficient sputum that passed quality control. We identified 10 overlap DEMs for the comparison between bronchiectasis patients and healthy subjects, and between PA and non-PA colonization group. Both miR-92b-5p and miR-223-3p could discriminate PA colonization (C-statistic >0.60) and independently correlated with PA colonization in multiple linear regression analysis. The differential expression of miR-92b-5p was validated by quantitative polymerase chain reaction (P <0.05), whereas the differential expression of miR-223 trended towards statistical significance (P =0.06). These DEMs, whose expression levels correlated significantly with sputum inflammatory biomarkers (interleukin-1β and interleukin-8) level, were implicated in the modulation of the nuclear factor-κB, phosphatidylinositol and longevity regulation pathways.
Conclusion: Sputum exosomal microRNAs are implicated in PA colonization in bronchiectasis, highlighting candidate targets for therapeutic interventions to mitigate the adverse impacts conferred by PA colonization.
Keywords: bronchiectasis, microRNA, exosome, Pseudomonas aeruginosa , biological pathway